Which Ions Were Present in the Original Solution
I believe the answer is NaCl but I am not sure and this precipitate only formed in the first NaOH extract as precipitate was essentially nonexistent in the 2nd extract. When you add sodium sulfate to the solution a white precipitate forms.

A Chromate Dichromate Equilibrium Experiment Rsc Education
6 months 2 weeks ago.
. Ag Ca 2 and Fe 2. Ag Ca2 and Cu2. When sodium chloride is added to the solution no precipitate occurs.
So if you have a compound that dissociates into cations and anions the minimum concentration of each of those two products will be equal to the concentration of the original compound. When you add sodium sulfate to the solution a white precipitate forms. The precipitate is filtered off and potassium sulfate is added to the remaining solution producing no precipitate.
When sodium chloride is added to the solution no precipitate forms. The precipitate is filtered off and sodium carbonate is added to the remaining solution producing a precipitate. When you add sodium sulfate to the solution a white precipitate forms.
MathrmAg mathrmCa2 and mathrmCu2 When you add sodium chloride to the solution no precipitate forms. Then the precipitate is filtered off and sodium carbonate is added to the remaining solution and a precipitate forms. Hg2 2Ba2 Fe2 and.
When potassium carbonate is added to the remaining solution a precipitate. A g C a 2 and C u 2. When potassium chloride is added to the solution a precipitate forms.
A solution contains one or more of the following ions. First the nanocomposite synthetic adsorbent containing Fe3O4 magnetic particles stabilized by cetyltrimethylammonium bromide CTAB was synthesized and then the sample. Ag Ca 2 and Cu 2.
A solution contains one or more of the following ions. When you add sodium chloride to the solution no precipitate forms. A solution contains one or more of the following ions.
The precipitate is filtered off and sodium carbonate is added to the remaining solution producing a precipitate. A solution contains one or more of the following ions. When potassium chloride is added to the solution a precipitate forms.
A solution contains one or more of the following ions. Part A Which ions were present in the original solution. You filter off the precipitate and add sodium carbonate to the remaining solution producing another precipitate.
A solution contains one or more of the following ions. Heres how that works. Write net ionic equations for the formation of each of the precipitates observed.
Ag Ca2 and Cu2. Find step-by-step Chemistry solutions and your answer to the following textbook question. The concentration of ions in solution depends on the mole ratio between the dissolved substance and the cations and anions it forms in solution.
When sodium sulfate is added to the solution a white precipitate occurs. A solution conta ins one or more of the following ions. When you add sodium sulfate to the solution a white precipitate forms.
We can determine the amount of moles of thiosulfate required to titrate x moles of Sodium Thiosulfate. Which ions were present in the original solution. Which ions were present in the original solution.
When sodium sulfate is added to the solution a white precipitate forms. Ag Ca2 and Cu2. When sodium sulfate is added to the solution a white precipitate forms.
When sodium chloride is added to the solution no precipitate forms. Ag Ca2 and Cu2. Chemistry questions and answers.
A solution contains one or more of the following ions. When you add sodium sulfate to the solution a white precipitate forms. Hg2 2 Ba2 and Fe2.
Find step-by-step Chemistry solutions and your answer to the following textbook question. Hg_2 2 Ba2 and Fe2. You filter off the precipitate and add sodium carbonate to the remaining solution producing another precipitate.
When you add sodium chloride to the solution no precipitate forms. July 2 2021 July 2 2021 thanh. A solution contains one or more of the following ions.
Ag Ca2 and Cu2. A solution contains one or more of the following ions. When sodium chloride is added to the solution no precipitate forms.
Ag Ba2 and Cu2. A solution contains one or more of the following ions. You filter off the precipitate and add sodium carbonate to the remaining solution producing another precipitate.
When you add sodium chloride to the solution no precipitate forms. Up to 10 cash back Abstract In the present study an adsorption method exploiting synthetic and natural adsorbents was utilized for removing cobalt ions from aqueous solutions. The precipitate is filtered off and potassium sulfate is added to the remaining solution producing no precipitate.
When potassium carbonate is added to the remaining solution a precipitate forms. When you add sodium chloride to the solution no precipitate forms. When you add potassium chloride to the solution a precipitate forms.
We can now determine the concentration of ClO- ion in the original bleaching solution by examining the volume of solution titrated the volumeconcentration of titration used and the total stoichiometry of equations 1 and 2. When sodium sulfate is added to the solution a white precipitate forms. When you add sodium chloride to the solution no precipitate forms.
A solution contains one or more of the following ions. When 250 mL of a solution containing both Fe2 and Fe3 ions is titrated with 230 mL of 00200 M KMnO4 in dilute sulfuric acid. A solution contains one or more of the following ions.

When You Dissolve Copper Chloride Into Water It Makes A Solution When You Put Pure Aluminum Foil Into The Solu Science Skills Chemical Energy What Is Science

Refining Palladium From 10 Pd C This Awesome Color Is Caused By The Aqua Regia And The Palladium Ions Organic Chemistry Chemistry Chemistry Labs

Lesson Explainer Further Tests For Anions Nagwa

Lesson Explainer Further Tests For Anions Nagwa

Calculating Ion Concentrations In Solution Youtube

Ion Concentration In Solutions From Molarity Chemistry Practice Problems Youtube
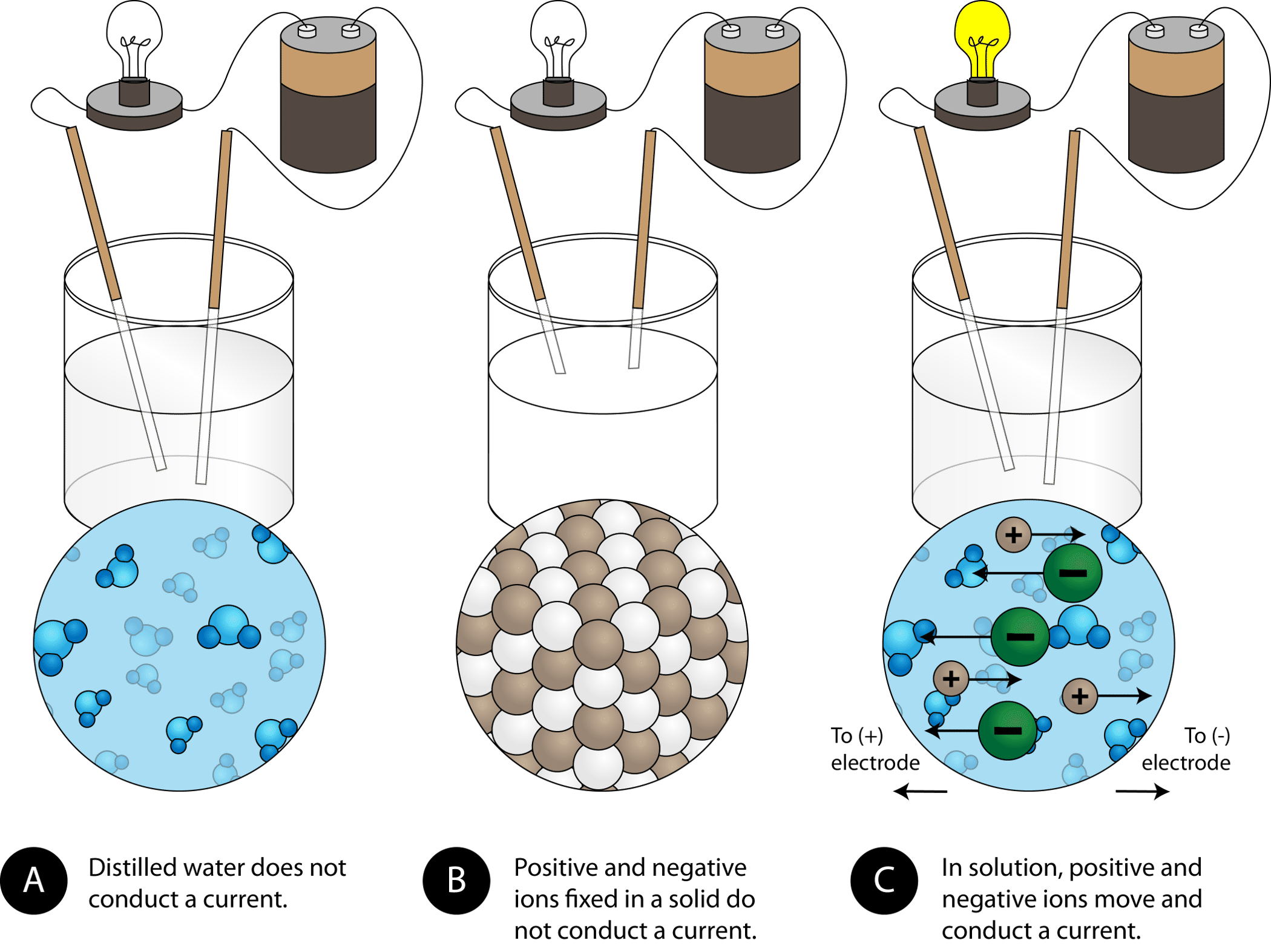
Physical Properties Of Ionic Compounds Ck 12 Foundation

This Is My Original Post Tumblr Littlepostitnotes Studying Inspo Study Inspiration Study Motivation
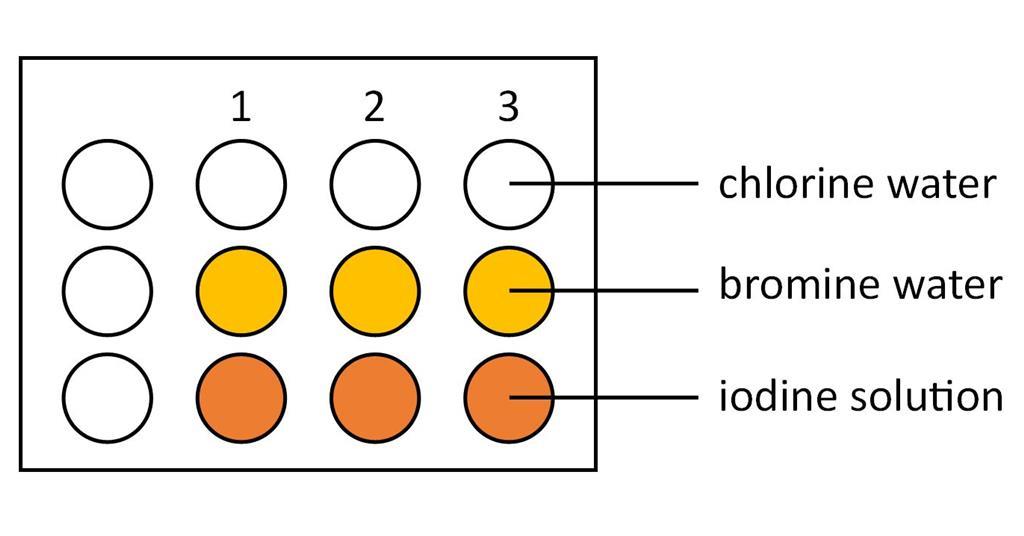
Halogens In Aqueous Solution And Their Displacement Reactions Experiment Rsc Education

Phillips Milk Of Magnesia Tablets With An Original By Coolvintage 14 50 Butylka
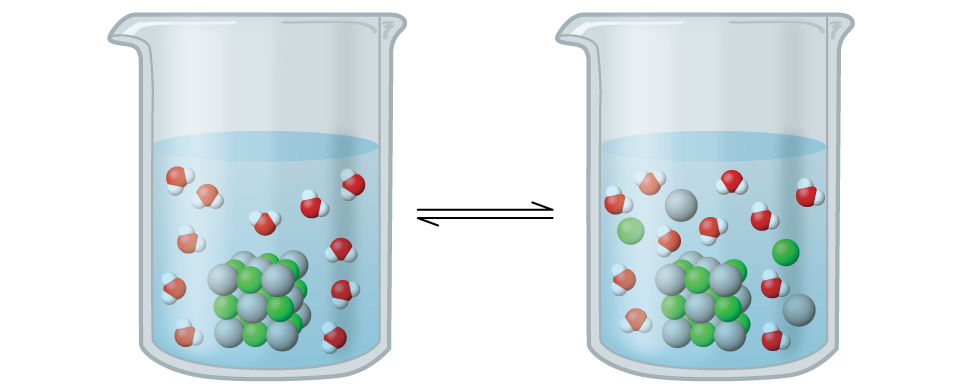
15 1 Precipitation And Dissolution Chemistry

Nomenclature Chart Chemistry Education Nomenclature Chemistry Chemistry

Lesson Explainer Further Tests For Anions Nagwa
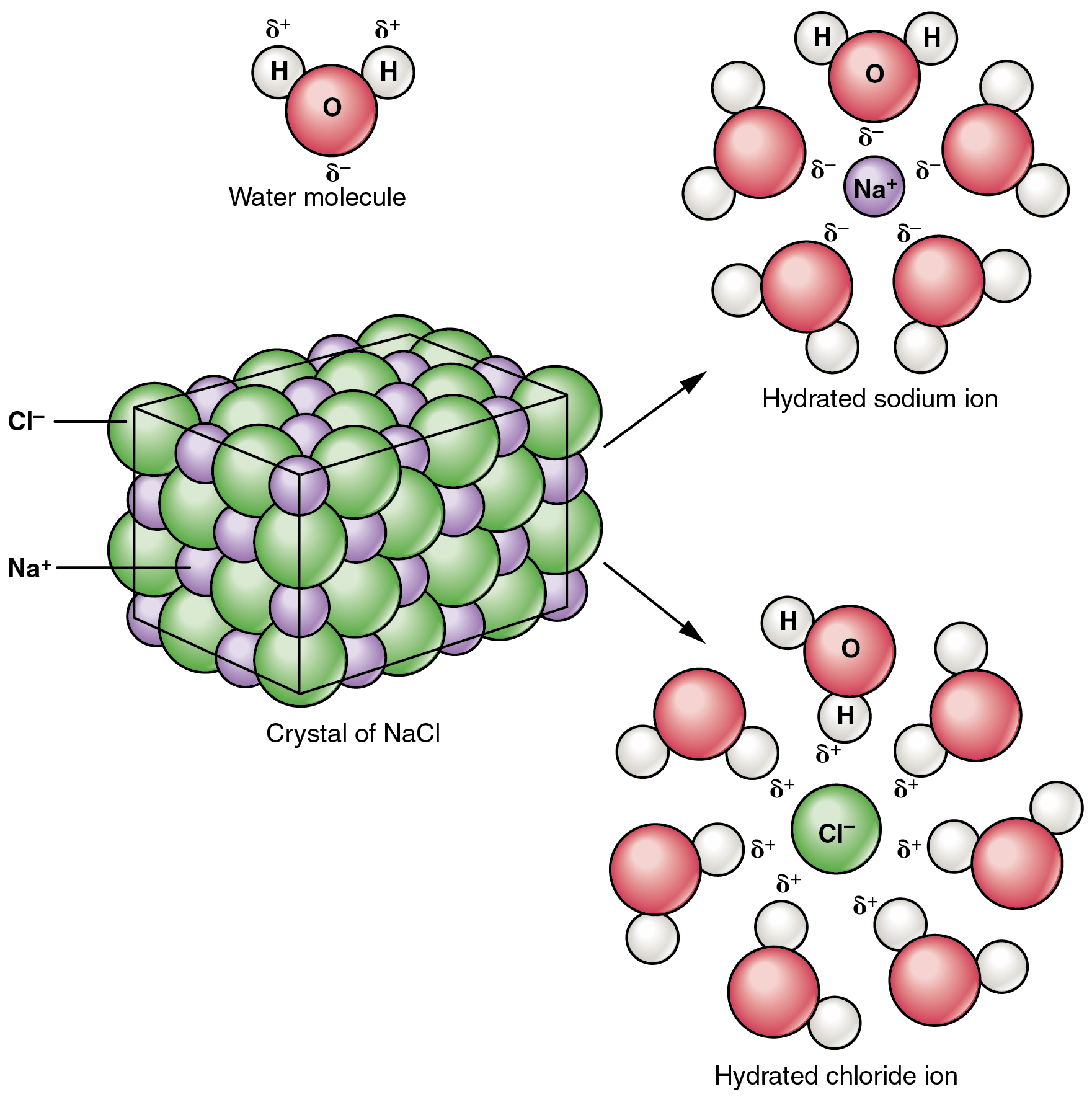
Molecular Complete Ionic And Net Ionic Equations Article Khan Academy
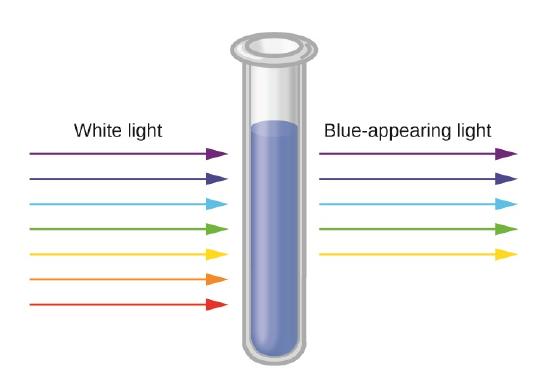
24 7 Color And The Colors Of Complexes Chemistry Libretexts
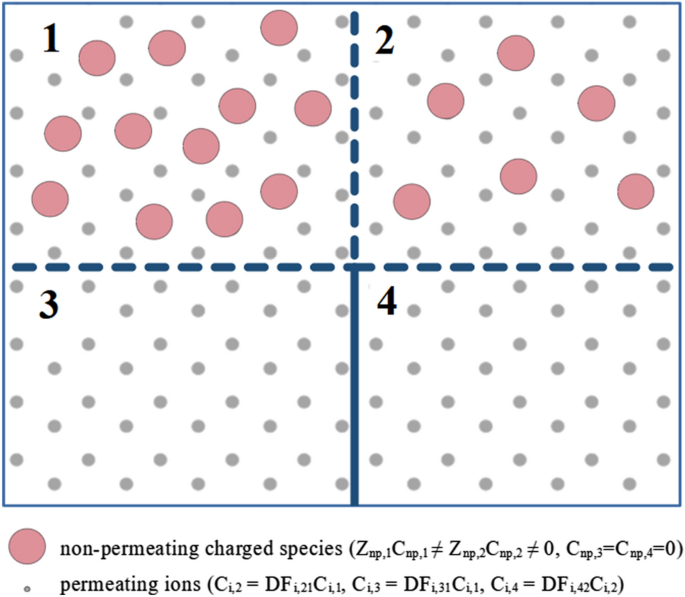
Calculation Of The Gibbs Donnan Factors For Multi Ion Solutions With Non Permeating Charge On Both Sides Of A Permselective Membrane Scientific Reports

Using Sodium Hydroxide Solution To Identify Metal Ions Video Lesson Transcript Study Com

Comments
Post a Comment